Can you tell the difference between effects from depression and antidepressants?
The swirl of daily emotions is hard enough for most of us to understand well. The addition of multifaceted, changing effects from any regular medication can make this an especially complex question.
Dear readers — this is one of the most important things I’ve ever published. With roots that date back to my 2006-2009 University of Illinois dissertation on depression treatment, I’ve been thinking about this question for two decades now.
I’m sharing below a new Antidepressant Side Effect Checklist that I’ve been working on based on the latest research for public educational purposes. Before introducing it, I need to say more about why this question matters so much.
There’s a lot to say before introducing an Antidepressant Side Effect Checklist, precisely because of how much depression and anxiety hurt. I’ll never forget a young woman in her 20s comparing her experience with depression to earlier, vicious child abuse. This is what she told me:
“I have had beatings to the point of unconsciousness — ripped, broken, arms taken out of the socket — and that compares nothing… doesn’t even begin to be the pain that became every day, just right here [pointing to chest] — this thing that wouldn’t go away — that made it hard to breathe… I would rather have hours and hours of people beating me than to have been where I was inside. It hurt that bad… there were times I thought it would kill me — all on its own.”
That’s how intense and overwhelming depression can become. And it’s why it makes sense that someone facing this would do anything they can to find relief, stability, and hope. Whatever path someone chooses in that effort deserves understanding and support. That includes deciding to take an antidepressant — for as long as it feels helpful or necessary.
The purpose of this inventory is simply to offer greater clarity about the range of possible side effects associated with antidepressant treatment, so that families and individuals — young and old — can participate more actively in decisions about their care.
For some, that may simply mean continuing treatment with more awareness. For others, it might raise questions worth exploring over time with a trusted professional. In some instances, that conversation may eventually lead to considering cautious, gradual changes — with appropriate support and at a measured, careful pace. For many, no changes may be needed; instead, this may simply put language to experiences or confirm that their current approach is working.
As Jon Kabat-Zinn has taught, until we can see clearly where we are, we don’t really know where to go next. In my experience, many people are unaware or confused about antidepressant side effects — with little understanding about whether a headache or brain fog or sexual problem could be associated with their treatment.
Even so, greater clarity about this all doesn’t force any particular decision — nor does it require urgency. Expanded awareness simply allows wiser, more grounded decisions, which may support deeper healing over time. Whatever adjustments are made can happen gradually, without pressure — especially avoiding sudden changes without guidance.
Hoping this checklist is helpful,
Yours, Jacob
What’s really coming from what?
Since publishing a recent cautionary story about inadvertent treatment effects, I’ve been thinking more about how difficult it can be to differentiate between symptoms arising from emotional struggles like depression and the effects of medications many people now take as a first-line treatment option.
That confusion can really make a difference. A friend told me recently about piercing headaches that wouldn’t go away — headaches that she hadn’t experienced until recent years. Even though this woman was educated and bright, she hadn’t seriously considered that the antidepressants she had started during this same period might be contributing to her migraines.
That specific side effect is listed on 5 out of 6 FDA prescribing guides of the most common antidepressants. But my friend likely never heard about that possibility from her physician — nor did she read about it on her own. If she had picked up the pamphlet inserted into the package, she would also have little way of knowing that the “official” list of side effects may not capture the full range of what can occur, especially over the long-term.
This is another reason for the confusion: so many of these side effects naturally change over time in ways that can be unpredictable. Amid this uncertainty, strong biases can exert themselves. Psychiatrist Peter Breggin cautioned for decades how easy it can be to assume something unpleasant is coming from an underlying emotional problem, while taking for granted that something positive or pleasant is connected to the medical treatment.
Despite this confusion, I believe there’s a strong way to ground ourselves in this discernment. It starts with getting clearer about the full scope of side effects associated with a particular treatment. From my own renewed curiosity, I decided to gather the side effects submitted in the FDA approval process for the 6 most common antidepressants in use: Prozac, Zoloft, Celexa, Lexapro, Paxil and Luvox. These are each part of a group of classic SSRIs (selective serotonin reuptake inhibitors) — which were developed to inhibit serotonin from being reabsorbed in the brain, in an attempt to make more of that neurotransmitter available for someone struggling with depression.1
Supplementing with other data from post-marketing studies and consumer/patient forums, I’ve pulled the side effect themes together into a draft of a new checklist to assist those wanting to get a better sense of whether parts of their experience may be influenced by this particular class of medications.
That could be good news, by the way. If symptoms like headaches, dizziness, insomnia, or irritability are partly medication-related, they may be more modifiable than they seem.
In the wake of realizations like this, I’ve known many people who decided with their supervising doctor to make some gentle, thoughtful adjustments in how they respond to depression, and subsequently came to find a different kind of life experience (here’s one example).
A short-term symptoms focus
Virtually all of the mental health surveys I’ve ever seen are focused on symptoms of a particular emotional disorder — in this case, centered on the “depressed mood” or “loss of interest/pleasure” in life which are one of two conditions required for a clinical diagnosis of depression. In addition, at least three of the following need to be present: concerning shifts in weight/appetite, sleep, energy, cognitive ability, lack of movement, “feelings of worthlessness” and “recurrent thoughts of death.”
As the classic DSM depression profile, this constellation of symptoms must be present over a 2-week period and represent a “change from previous functioning.”
It’s here that our collective attention has been focused for nearly 3 decades: running through these criteria with a doctor, screening our young people, or taking a self-assessment ourselves. Often prompted by these kinds of assessments, an estimated 33–35 million Americans now take antidepressants annually (~13% of the adult population), along with roughly a million teenagers (~5% of the teenage total in the U.S.).
Given the vast scope of this public health intervention, it’s important to explore what happens next: understanding in greater detail the evolving range of effects people experience on the medications.
Yet after the investment in initial short-term studies designed for FDA approval, it’s striking how little sustained research has gone into careful evaluations of treatment experience over time, especially not in the long term. (I know, because over the years I’ve been collecting every long-term antidepressant study I can find).
For someone whose antidepressant isn’t going well over time, this can create some added confusion. That’s where one of my own dear family members was years ago. Medicated for depression, but feeling worse than ever.
After years of treatment, she was told that she was “treatment resistant.” Rather than looking more carefully at the treatment itself, she came away feeling that she was the problem — ‘guess I’m just even sicker than I thought?’
Another kind of symptom focus
We need to do better than that for the precious lives of so many amazing people facing emotional challenges. (In my experience it’s especially sensitive and deep souls who tend to hurt like this — Homer Simpson doesn’t get depressed).
For the many people — young and old — who yearn for a little more peace and happiness, I wonder: What could it mean if a little more attention went to appraising the full scope of antidepressant side effects potentially showing up in their experience?
For one, I believe we’d be closer to being able to see clearly what’s-coming-from-what in terms of our evolving emotional experience: Where is the pain coming from? And the sweetness too?
Just as that clarity could make a difference in important decisions about long-term trajectories of care, a lack of clarity on this question can surely complicate these same deliberations.
That’s my motivation in drafting this new “awareness tool” I’m laying the groundwork for introducing here. I hope this can be something to help suffering individuals or their loved ones if antidepressants have been a part of their recent life experience.
Illustrations generated with AI (ChatGPT), based on author direction
Some background
Twenty-five years ago during my doctoral program at the University of Illinois, I became fascinated with competing stories human beings tell about emotional pain — and the different ways they interpret and “narrate” depression, in particular. After interviewing people in Salt Lake City and Chicago for my dissertation on Prozac, I wrote 300 pages on the topic, including peer-reviewed articles on competing stories about the depressed brain, about whether recovery is possible and about what it means for medication to “work” for teens and children.
I later helped create two different online classes drawing on many of these same concepts and wrote a 2023 Deseret News article, “Is it me or my medication,” on this specific question I’m taking up today in more depth.
One of the biggest things I learned in all my research on the topic is how complicated this is all — not easy to tell what’s going on! Once someone is taking a daily medication that directly influences their emotion, a 3rd variable gets introduced that isn’t so easy to parse out. After all, whatever you’re experiencing inside is just what you are experiencing … how is someone supposed to tell whether it’s connected to medication or any of the hundreds of other (environmental, lifestyle, social, physical, spiritual) influences on their emotions?
It was the Boston College sociologist David Karp who first gave this question proper attention in his memoir of grappling with depression himself: “Is it me or my meds?”
My own study reinforced how challenging this question is for so many people. Yet outside of passing conversations, I’ve never had a chance to create anything specifically for people in the middle of this question.
What follows is shared as public scholarship to inform further conversations within families and the many helping support those facing these emotional challenges. It represents a synthesis of existing knowledge, and I welcome additional critique and feedback.
The development approach I took
Here’s how I created this antidepressant side effect checklist.
Understanding the basic scope of side effects
To understand the scope of side effects of any medication, a good place to start is the Full Prescribing Information (FPI) — an FDA-approved “package insert” for healthcare providers that compiles a drug’s safety profile based on data reported by the manufacturer.
After gathering both common and serious side effects in FPIs for the 6 most established SSRI antidepressants2 — Prozac, Zoloft, Celexa, Lexapro, Paxil and Luvox — I identified 22 different side effects that showed up across all six medications, along with another 7 that showed up in at least 5 of the medications.
Here is the 6-out-of-6 list of potential side effects (with some medical jargon converted to plain terminology):
Feeling like you might throw up
Diarrhea or loose stool
Difficulty having bowel movements
Indigestion or upset stomach
Dry mouth
Trouble sleeping
Feeling very sleepy or drowsy
Low energy or unusual tiredness
Dizziness or lightheadedness
Shaking, usually in the hands
Feeling anxious or worried
Feeling nervous or jittery
Excessive sweating
Not feeling like eating
Decreased sex drive
Delayed or difficulty with ejaculation
Thoughts about self-harm or suicide
Feeling suddenly much worse emotionally or more depressed
Feeling unusually energized, impulsive, or out of control
Unusual bleeding or bruising
And here is the 5-out-of-6 side effect list, based on these same six FPI guides:
Stomach pain
Throwing up
Feeling restless or unable to relax
Difficulty getting or maintaining an erection
Severe restlessness or inability to sit still
Symptoms of too much serotonin (confusion, sweating, fast heartbeat, shaking)
Eye pain or sudden vision changes
Headache
Difficulty reaching orgasm
Skin rash
As you can see, many potential SSRI side effects are not unique to individual drugs — they are shared across this class of medications. These SSRI side effects that have been identified as potentially happening in all six medications include gastrointestinal distress, sleep and energy disruption, neurological symptoms like dizziness and tremor, heightened anxiety or restlessness, sexual dysfunction, and a smaller but important set of serious risks affecting mood, bleeding, and overall system regulation.
This constellation of experiences forms a natural anchor point for any antidepressant side effect inventory. In fact, approximately 85% of the items in two of the main antidepressant symptom inventories already in use today map onto this same basic list of symptoms above: The 32-question Toronto Side Effects Scale (TSES) from 2002 and the 21-question Antidepressant Side-Effect Checklist (ASEC) from 2009 both largely draw from the same clusters of symptoms highlighted in FDA prescribing documents.
This only becomes a limitation if additional categories of side effects are not considered. For instance, neither of these earlier side-effect surveys explore the possibility of an antidepressant worsening depression, increasing suicidal thoughts, inducing mania, or prompting symptoms of “serotonin syndrome” (signs of too much serotonin). And like the FPI prescribing brochures themselves, neither of these other surveys considers the possibility of a drug-induced increased sense of hopelessness, worthlessness or loss of meaning.
Balancing side-effects with positive effects
Of course, antidepressant treatment also frequently elicit positive effects. In line with research submitted to the FDA for antidepressant approval, many people describe a reduction in depression symptoms (and sometimes in anxiety symptoms) that makes any of these known side effects potentially worth tolerating in an overall cost-benefit ratio.
That means lower scores on depression scales like the Hamilton Depression Rating Scale, with some modest improvements in sleep, appetite and energy also seen over 4–8 weeks among roughly half of patients during early weeks on an antidepressant, and a “remission” observed among many of these symptoms in about a third of patients during this same initial period.
These patients describe things like:
“Relief from the worst intensity” for a period of time and feeling less overwhelmed with the pain becoming more manageable (“taking the edge off”).
A better ability to get out of bed, work and engage in daily tasks, as well as improved sleep for some.
Less constant worry or panic, and feeling more settled or calm
Fewer extreme highs/lows and more predictable mood
Sometimes an increase in motivation and a return of interest in life events
I heard these same kinds of reports in my University of Illinois antidepressant study, especially during the initial period of treatment. For most people I’ve spoken with, unfortunately, these benefits do not endure in the same way over time — underscoring the importance of longer-term research.
These initial positive effects have received enormous attention over the years — publicized and promoted for nearly two decades. By comparison, details on side effects have received relatively sparse public attention.
All this explains why more discussion is needed and, in my view, something more than earlier side effect inventories, which largely follow company-reported lists of symptoms.
Two kinds of additional data
There are two kinds of additional information important to consider in understanding the range of side effects on any medication.
The value of independent research
When side-effect surveys rely primarily on company-generated data, an important set of additional possibilities will likely be missed. This is intuitive to anyone who has watched a drug commercial with lovely video images, accompanied by a rapid voiceover running through a list of side effects.
It turns out even the list of side effects companies insert via pamphlet into a medication box is curated in interesting ways. For example, sexual side effects such as reduced libido, delayed ejaculation, erectile dysfunction, or difficulty reaching orgasm, are compressed into a single, more generic label of “sexual problems” in the official medication guides for Celexa and Lexapro.
To capture the wider picture of side effects, these company reports need to be supplemented with “non-official” sources. In addition to independent outcome studies, there is value in studying consumer/patient forums without any connection to industry or professional associations.
The value of patient forums
Consumer/patient forums can be uniquely helpful in identifying what might be missing from official symptom lists. While official sources — company and independent — define what is identified in research, patient forums reveal what is experienced.
While clinical trials capture frequent, measurable, short-term effects and physiologically clear outcomes, patient forums also do a better job capturing subjective experience, cognitive and emotional changes, identity shifts, earlier onset symptoms, changes over time (including long-term effects and withdrawal effects), or changes that are rare or spread out over time, and thus harder to pinpoint.
During her time on faculty at Colorado State University, my colleague Shannon Hughes joined David Cohen in comparing antidepressant side-effect reports on industry-funded websites (like www.webmd.com), with independent patient forums (like www.askapatient.com), which they found “capture real-world variability missing from trials.”
By comparing Lexapro side effects presented on these two different kinds of websites, they found:
Professionally-controlled websites were more likely to feature symptom improvement, while consumer-controlled sites tended to share more details about symptoms worsening.
Consumer sites were also more likely to share side effects that were “disrupting daily routines” and which involved “richer descriptions of effects in context.”
These researchers concluded: “Professional medication descriptions offer the advantage of a concise yet comprehensive listing of drug effects, while consumer reviews offer greater context and situational examples of how effects may manifest in various combinations and to varying degrees.”
Additional themes documented
As you can see, if we want to better understand the emotional, cognitive, interpersonal, and sensory experiences that most people consider central, we need to step beyond official side-effect lists to explore the broader picture of lived experience on any given medication.
Based on that broader analysis, here are some other potential antidepressant side effects that are commonly observed in patient forums and independent research:
Emotional numbness or blunting
Feeling less emotional range (less joy and less sadness)
Feeling less awe, wonder, or being “moved” by meaningful experiences
Reduced motivation or drive
More apathy or less interest in things you usually care about
Brain fog (feeling mentally dull or less sharp)
Memory problems
Reduced empathy or compassion for others
Feeling more emotionally distant or disconnected from other people
Less interested in being around others
Inner restlessness or inability to sit still
Extreme internal agitation and urge to move (“feel like I’m crawling out of my skin,” sometimes called akathisia)
Internal vibrations or buzzing sensations
Muscle twitching or jerking
Ringing in the ears (tinnitus)
Vivid dreams or nightmares
Night sweats
Temperature sensitivity (feeling unusually hot or cold)
Jaw clenching or jaw tightness
These are all underrepresented in FDA “pamphlet inserts” or FPI “clinician guides.” I’ve included them in this new checklist below.
Two other areas of inquiry
In addition to this wider scope of side effects captured in patient forums and independent research, there are two other areas that make this checklist unique, summarized below.
1. Other kinds of non-SSRI antidepressants
After forming the original side effect list from the 6 most popular, SSRI antidepressants (Prozac, Zoloft, Celexa, Lexapro, Paxil and Luvox), I realized this list of potential changes mapped fairly well onto what’s known about other antidepressants too, with a few exceptions.
In order to make this checklist more generalizable to Wellbutrin and some newer antidepressants like Trintellix, and SNRIs like Pristiq or Cymbalta, I’ve added several potential side effects often associated with these newer medications:
Increased appetite
Weight gain
Feeling unusually sleepy, sedated, or hard to wake up
Feeling unusually activated, stimulated, or “amped up”
Heart racing, pounding, or palpitations
Unusual increase in sexual thoughts, desire, or arousal
Mood swings or feeling emotionally unstable
Blurred vision or other visual changes
Changes in urination (more frequent, difficult, or unusual)
2. Withdrawal-specific changes
While many of these side effects show up in the normal course of antidepressant treatment (e.g., weight gain, sexual dysfunction), a surprising number occur especially when making changes to the medication, whether that is starting, stopping, or adjusting dosage.
These dosage-change-specific experiences include some of the most common antidepressant side effects like headache, nausea, loss of appetite, agitation, difficulty concentrating, insomnia, fatigue and restlessness.
But there’s a third category of effects exclusive to withdrawal or discontinuation — and not typically showing up unless someone attempts to come off their medication (especially if they do so too quickly, and without tapering gradually enough).
Many of the antidepressant side effects that occur during initial treatment — especially in sleep, mood, energy, and anxiety — can also reappear during withdrawal, often with similar intensity. A core list of changes that are specific to withdrawal have been included in a separate section of the checklist:
Brain zaps / electric shock sensations
Internal buzzing or vibrating sensations
Dizziness triggered by head or eye movement
Feeling off-balance or like you’re walking on a boat
Sensitivity to light, sound, or motion
Sudden emotional surges or swings
Feeling detached from yourself or surroundings (unreal or distant)
Visual disturbances (e.g., trailing images or delayed visual tracking)
Symptoms that come in sudden waves or bursts
Symptoms that worsen quickly when a dose is missed and improve after taking it
This smaller list of potential withdrawal effects represents a broader group of possible effects, with 187 withdrawal-related symptoms that have been observed in people discontinuing psychiatric medication generally, as compiled here by former patients.
Read this first
Before going further, it’s worth pausing for a moment.
This checklist is not a diagnostic or treatment tool, and nothing here is intended as medical advice. That means this isn’t a clinical tool designed to diagnose, assess, or guide treatment decisions on its own. Rather, it is intended as an educational and reflective resource — to help people notice patterns and generate questions they may wish to explore with qualified professionals, similar to many other self-guided mental health checklists out there.
Most of what’s included here is already known in different places — clinical documents, studies, and patient reports — but rarely brought together in one accessible format. As a synthesis of publicly available sources, clinical documents, and patient-reported experience that is translated into a practical awareness tool, my aim is to make existing knowledge more visible and usable to the general public.
The final list is intentionally broad — gathering many possibilities. Yet most people will experience only a small fraction of these, and often temporarily. The goal here is not to alarm, but to give language to experiences that can otherwise feel confusing or isolating. If something stands out, it’s simply an invitation to notice — not to act quickly or make changes on your own.
People’s experiences with medication vary widely, and no single pattern applies to everyone. For many people, two things can be true at once: a medication may provide meaningful relief while also introducing side effects that are easy to miss. This complexity is part of what makes clear awareness so challenging.
The complete checklist
You can access here the full 90-question Antidepressant Side Effect Checklist, along with a reflection section with some questions as you consider the results.
I’ve worked hard to use language and categories that are accessible and reflective of how individuals actually experience these side effects — minimizing medical jargon.
While other nuanced questions could be asked about each item, including whether a particular symptom was “Mild,” “Moderate” or “Severe” (or not present at all), I’ve made each question simply a “yes” or “no” to reduce the cognitive load and make the checklist more doable. (I’d welcome feedback on the checklist and how I can make it better: jzhess@gmail.com).
None of this, of course, substitutes for professional medical advice. If anything in this list raises concern, the next step is not to make any changes on your own, but to begin new conversations with people you trust, including qualified professionals. Rapid changes — especially stopping suddenly — can make symptoms significantly worse.
That’s why it’s strongly advisable to work with a trusted medical professional in supervising decisions related to medication. And virtually always (unless you’ve only been taking a medication for a few months) — even if your doctor is recommending that you “cut the dose in half” — a decrease in medication often discussed in terms of gradual, stepwise reductions (for example, small percentage decreases over time)3
This checklist expands the scope of antidepressant effects worth considering, compared with both standard side-effect lists in the FDA Medication guides/FPI documents and earlier inventories that are more narrowly focused. For instance, in addition to other inventories reviewed earlier, American psychiatrist John Rush and colleagues developed the Patient Rated Inventory of Side Effects (PRISE) in the early 2000s as part of the STAR*D research project. What started off as only 8 questions is now a 32-question survey — but is not publicly available, to my knowledge.
Many of these inventories are either two decades old or simply too limited in the scope of side effects they take up. Other tools like the UKU Side Effect Rating Scale were developed by Scandinavian researchers to be more comprehensive across all psychiatric side effects. But again, this was created in 1987 and is now almost as old as I am!4
Two follow-up questions
There’s a lot to consider here. Let’s walk through two other questions some people will have.
1. Okay, but how frequent are any of these side effects?
To see a checklist of all potential side effects in one place can feel overwhelming. And it’s important to address a significant misconception about the true frequency of any given effect.
Please remember, especially, that this checklist aggregates possible symptoms that can emerge (or not emerge) from antidepressant treatment. And when a side effect does emerge, it may often only emerge for a short time.
Other side effects, of course, can be for a longer duration too.
This is important to keep front of mind so this antidepressant side effect checklist is seen for what it is (“these might show up”) — and not over-interpreted as something it’s not (“this is what someone is going to experience on SSRI antidepressants”).
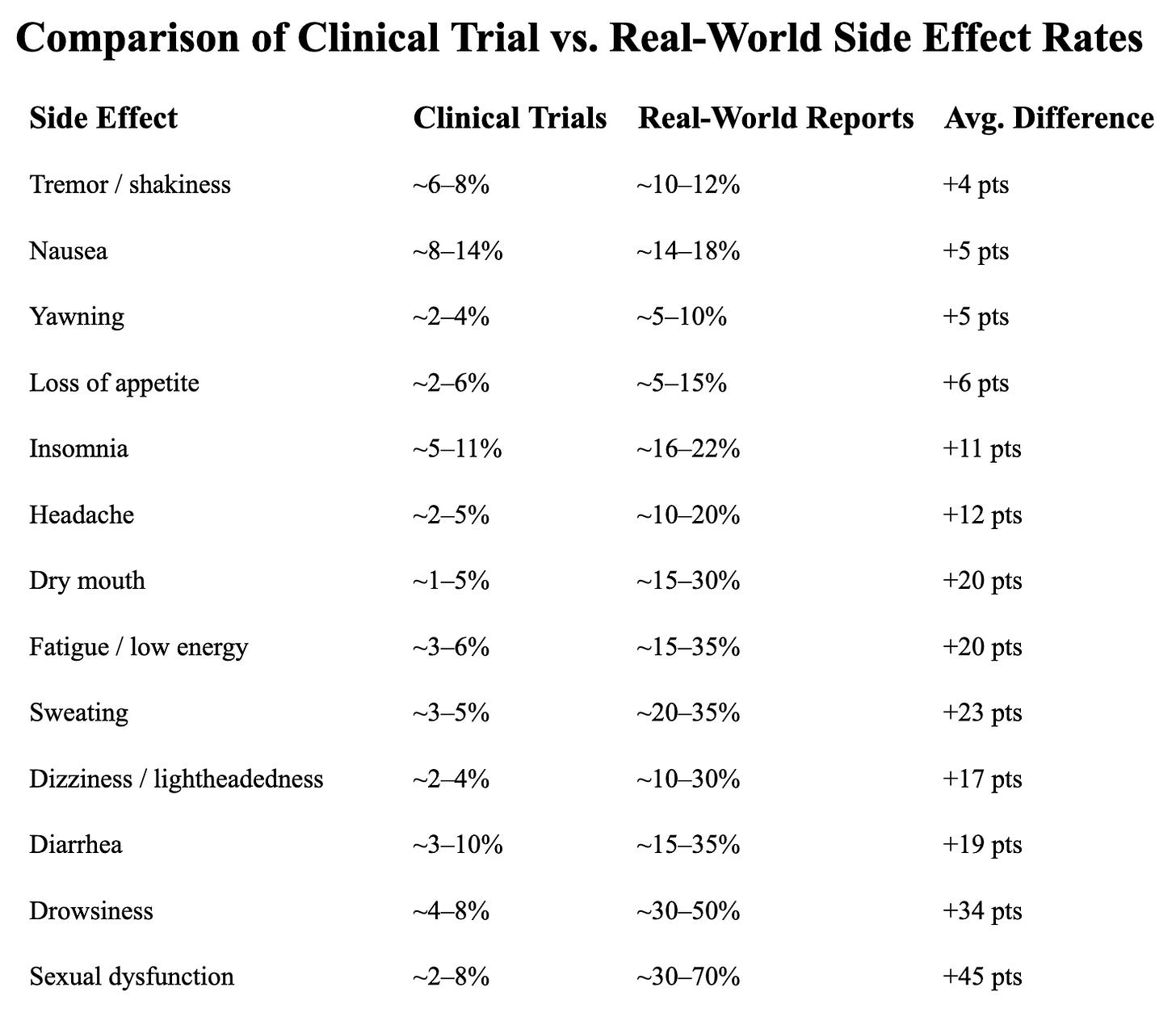
It’s well-known, of course, that side effects vary in frequency. What’s been less appreciated is how different estimates of these frequencies also vary considerably. For example, rates reported in clinical trials — often based on short durations and spontaneous reporting (“if you notice something happening, let the researcher know, okay?”) — tend to be lower, while studies using structured assessments or longer-term follow-up tend to find higher prevalence for various side effects.
For example, estimates of sexual dysfunction accompanying antidepressants range from 2-8% in company trial data, while more systematic research finds rates as high as 70%. Although that gap is especially dramatic, the difference between reported side effects observed in clinical trials and rates reported in real-world experience is consistently significant, as illustrated by this table below (ordered from smallest to largest difference):
As you can see, while some side effects — like tremors, nausea5, yawning and appetite changes — show somewhat similar rates across clinical trials and real-world reports, others differ substantially.6
2. Does this apply to teens?
A majority of these side effects in the checklist can occur in both adults and teens, who experience most of the same medication-related effects as older adults. That includes the core symptoms most commonly experienced: GI issues (nausea, appetite), sleep changes, fatigue, anxiety / agitation, and neurologic symptoms (tremor, restlessness).
Sexual side effects can also occur in teens, even those who are not sexually active. Some effects — especially restlessness, emotional changes, or disturbing thoughts — may be more noticeable and especially important to monitor in younger individuals. Teens also experience higher sensitivity to activation — meaning, they are more likely to experience agitation, restlessness, feeling “sped up” and behaviorally disinhibited.
This might explain why a risk of suicidal thoughts and emotional volatility is also higher for teens taking antidepressants (hence, the black box warning). This is especially true during initial treatment or when dosage is changed.
This may also explain why emotional and identity effects may be stronger for teens — e.g., feeling “not like myself,” emotional blunting and social/relational changes.
Two areas not included in the checklist
This tool does not attempt to be all things for all people, with two areas I’ve held off in including for now.
1. Pregnancy-specific changes.
There are several pregnancy-related adverse changes that can happen in expectant mothers taking antidepressants, as documented by independent studies and patient/consumer websites. Those include increased risk of postpartum bleeding, preterm birth (delivery before 37 weeks), miscarriage (also called “spontaneous abortion” in some studies), lower birth weight, and temporary newborn symptoms (irritability, jitteriness, feeding/sleep issues, mild respiratory distress).
Heart defects can also unfortunately occur, especially with Paxil in the first trimester, which resulted in a $1 billion settlement in 2010 that GlaxoSmithKline paid involving hundreds of affected families.
I’ve chosen not to include those in this checklist, because of its current length and because they would not apply to most people taking the survey. Here are the ten questions that could be added for the subset of women for whom they would apply:
Pregnancy-related changes
If you were pregnant while taking an antidepressant, or shortly after, did you notice any of the following? Note: these experiences can have many causes, including underlying health conditions or pregnancy-related factors.
Heavier than expected bleeding during or after delivery (postpartum bleeding) ☐ Yes ☐ No
Delivery earlier than expected (before 37 weeks) ☐ Yes ☐ No
Lower birth weight than expected ☐ Yes ☐ No
Pregnancy loss (miscarriage) ☐ Yes ☐ No
Newborn irritability, jitteriness, or unusual fussiness ☐ Yes ☐ No
Low blood sugar, low body temperature, or poor muscle tone in the newborn period ☐ Yes ☐ No
Feeding difficulties in the newborn period ☐ Yes ☐ No
Sleep difficulties or unusual sleep patterns in the newborn ☐ Yes ☐ No
Breathing difficulties or need for extra monitoring after birth ☐ Yes ☐ No
A diagnosed heart condition in the baby (especially if exposure occurred early in pregnancy) ☐ Yes ☐ No
2. Long-term changes
The majority of items represented in the checklist reflect the evidence arising from short-term research upon which FDA review is based. Drawing on the longer-term antidepressant outcome research available, another section could be added that addresses other changes over time in how people’s mood, recovery, and treatment experience evolve across years.
I’ve opted to not include these questions in the checklist as well, since there is relatively little known about the full extent of how treatment and biological effects evolve over time. But based on what we do know from available research, these questions below represent an accurate glimpse, generally speaking, of the longer-term trajectory of symptom and side effect evolution many people experience over time.
Long-term changes
Some people also notice longer-term changes in how their mood, recovery, and emotional experience evolve over time. Once again, these patterns may also be influenced by many other circumstances across a life trajectory.
Depressive episodes becoming more frequent or returning more quickly than before ☐ Yes ☐ No
Depressive periods lasting longer than in the past or feeling less able to return to your previous level of well-being after an episode ☐ Yes ☐ No
A more persistent, harder-to-change low mood (including between episodes) ☐ Yes ☐ No
Feeling more sensitive to stress or more easily triggered into low mood than before ☐ Yes ☐ No
Emotional range becoming more limited over time (even if initially helpful) ☐ Yes ☐ No
Feeling stuck at a partial level of improvement or that your emotional baseline has shifted in a way that feels harder to change ☐ Yes ☐ No
Feeling like your mood or functioning increasingly depends on continuing the medication ☐ Yes ☐ No
Medication becoming less effective over time (e.g., needing higher doses or initial benefits like relief, motivation, or emotional lift fading) ☐ Yes ☐ No
Still not so easy to tell!
With any serious exploration of antidepressant side effects, humility is called for — for many reasons. First of all, the effect of any particular medication is expected to change over time, especially with dosage adjustments.
Second, our emotions can be naturally influenced from many different sources. And it’s naturally easy, when reading a list like this, to see oneself in many items. But that doesn’t mean all the changes apply or are medication-related.
Depression itself, as highlighted above, can also generate many difficult emotional effects. This creates a three-way ambiguity, with a given symptom potentially arising from the condition, the treatment, or the process of coming off the treatment.
Bottom line: even when an association exists, it’s difficult to say for sure what arises from what.
Obviously, correlation is not causation. Yet that doesn’t dismiss the value of correlations, which can teach us a lot — as long as we approach them in the right light.
Similar to the FIBSER (Frequency, Intensity, Burden of Side Effects Rating), we might also ask: how confident should any of us feel about whether a particular change is connected to antidepressant treatment, compared with something else?
One of the clearest ways to tell whether a medication has been contributing to any of these side effects is to make an adjustment (with appropriate supervision), and then observe whether the specific side effect changes — asking specifically:
Does the effect get worse after a dosage increase?
Does it improve after a dosage decrease or tapering?
These are challenging questions. So, let’s be humble. But we should also be serious. If a medication were making life harder, would we recognize it?
Above all, remember that awareness doesn’t require urgency. It simply opens the door to more informed and thoughtful decisions — made at the right pace, with the right support.
Notes:
Much remains unknown about the effects of introducing a SSRI in the brain, especially over time, as the late Elliot Valenstein at the University of Michigan, pointed out before his death. One thing neuroscientists do know is that the presence of a SSRI initially leads natural serotonin receptors in the brain to shut down, in order to preserve homeostasis in the brain. In order to avoid too much serotonin, these natural serotonin receptors end up “down-regulating.”
These side-effects identified in the FPI documents became my starting point:
Fluoxetine (Prozac): common adverse reactions listed in the FPI include headache, asthenia, flu syndrome, fever, nausea, diarrhea, anorexia, dry mouth, dyspepsia, constipation, flatulence, vomiting, weight loss, insomnia, nervousness, anxiety, somnolence, dizziness, tremor, decreased libido, abnormal thinking, yawning, sweating, rash, pruritus, and abnormal vision; serious warnings include suicidal thoughts and behaviors, serotonin syndrome, allergic reactions/rash, activation of mania or hypomania, seizures, altered appetite and weight, increased risk of bleeding, angle-closure glaucoma, hyponatremia, anxiety/insomnia, QT prolongation, cognitive/motor impairment, and sexual dysfunction.
Sertraline (Zoloft): common adverse reactions include nausea, diarrhea/loose stool, tremor, dyspepsia, decreased appetite, hyperhidrosis, ejaculation failure, decreased libido, somnolence, insomnia, agitation, constipation, fatigue, dry mouth, dizziness, abdominal pain, and malaise; serious warnings include suicidal thoughts and behaviors, serotonin syndrome, increased risk of bleeding, activation of mania or hypomania, discontinuation syndrome, seizures, angle-closure glaucoma, hyponatremia, QTc prolongation, false-positive benzodiazepine screening, and sexual dysfunction.
Citalopram (Celexa): common adverse reactions include nausea, diarrhea, dyspepsia, vomiting, abdominal pain, dry mouth, increased sweating, somnolence, insomnia, anxiety, anorexia, agitation, dysmenorrhea, decreased libido, yawning, tremor, ejaculation disorder, impotence, upper respiratory tract infection, rhinitis, sinusitis, fatigue, fever, arthralgia, and myalgia; serious warnings include suicidal thoughts and behaviors, QT prolongation and torsade de pointes, serotonin syndrome, increased risk of bleeding, activation of mania or hypomania, discontinuation syndrome, seizures, angle-closure glaucoma, hyponatremia, and sexual dysfunction.
Escitalopram (Lexapro): common adverse reactions include insomnia, ejaculation disorder, nausea, increased sweating, fatigue, somnolence, dry mouth, dizziness, diarrhea, constipation, indigestion, abdominal pain, influenza-like symptoms, decreased appetite, decreased libido, rhinitis, sinusitis, impotence, and anorgasmia; serious warnings include suicidal thoughts and behaviors, serotonin syndrome, discontinuation syndrome, seizures, activation of mania or hypomania, hyponatremia, increased risk of bleeding, angle-closure glaucoma, cognitive/motor impairment, use in patients with concomitant illness, and sexual dysfunction.
Paroxetine (Paxil): common adverse reactions include asthenia, sweating, nausea, decreased appetite, somnolence, dizziness, insomnia, tremor, nervousness, dry mouth, constipation, decreased libido, abnormal ejaculation/ejaculatory disturbance, impotence, female genital disorders, yawning, diarrhea, infection, headache, palpitation, vasodilation, rash, flatulence, dyspepsia, anxiety, and paresthesia; serious warnings include suicidal thoughts and behaviors, serotonin syndrome, reduced tamoxifen effectiveness, abnormal bleeding, angle-closure glaucoma, hyponatremia, bone fracture, activation of mania or hypomania, seizures, akathisia, cognitive/motor impairment, and sexual dysfunction.
Fluvoxamine (Luvox): common adverse reactions include nausea, somnolence, insomnia, asthenia, nervousness, dyspepsia, abnormal ejaculation, sweating, anorexia, tremor, vomiting, and in some analyses also anorgasmia, decreased libido, dry mouth, rhinitis, taste perversion, urinary frequency, agitation, depression, flatulence, hyperkinesia, and rash; serious warnings include suicidality, serotonin syndrome, angle-closure glaucoma, discontinuation reactions, abnormal bleeding, activation of mania or hypomania, and major drug-interaction warnings involving several contraindicated or high-risk concomitant drugs.
Inner Compass provides helpful public guidance on tapering best practice, led by a trusted colleague who experienced this tapering process in her own life and has helped thousands of people navigate these questions for a decade.
It also seems important to include some side-effects that show up less consistently so the instrument does not quietly erase real experiences just because they show up less uniformly across medications. In this case, that means including in the checklist potential side effects showing up in four of the six FPI guides for Prozac, Zoloft, Celexa, Lexapro, Paxil and Luvox as potential common or serious side effects:
Gas or bloating
Frequent yawning
Tingling or “pins and needles” sensations
Weight loss
Sexual difficulties in women
Runny or stuffy nose
Sinus congestion or infection
Itchy skin
General feeling of being unwell
Fever
Flu-like symptoms
Muscle aches or soreness
Very low sodium symptoms (confusion, headache, weakness)
Seizures
And potential side effects showing up in three of the six FPI guides:
Trouble thinking clearly or mental fog
Joint pain
Painful menstrual cramps
Cold-like symptoms
Fainting or heart rhythm problems
Severe allergic reaction (swelling, trouble breathing)
So, in other words, clinical trials suggest that antidepressants tend to increase nausea by about 8–14% above baseline (placebo), while independent reports show that roughly 14–18% of people report nausea they attribute to the medication. To summarize:
Clinical trials: ~8–14% increase in nausea above placebo
Real-world reports: ~14–18% report medication-related nausea
These ranges reflect synthesized estimates across clinical trial tables and multiple independent studies, rather than coming from a single source. Clinical trial estimates are also derived from short-term, placebo-controlled studies, while real-world estimates come from patient reports and observational research using different methods. Thus, these figures are not directly equivalent, but offer a useful comparison of what is measured in each context.